TRILLIANT HEALTH RESEARCH
Research
Timely trend analysis. Free in your inbox.
Receive weekly studies from Trilliant Health.
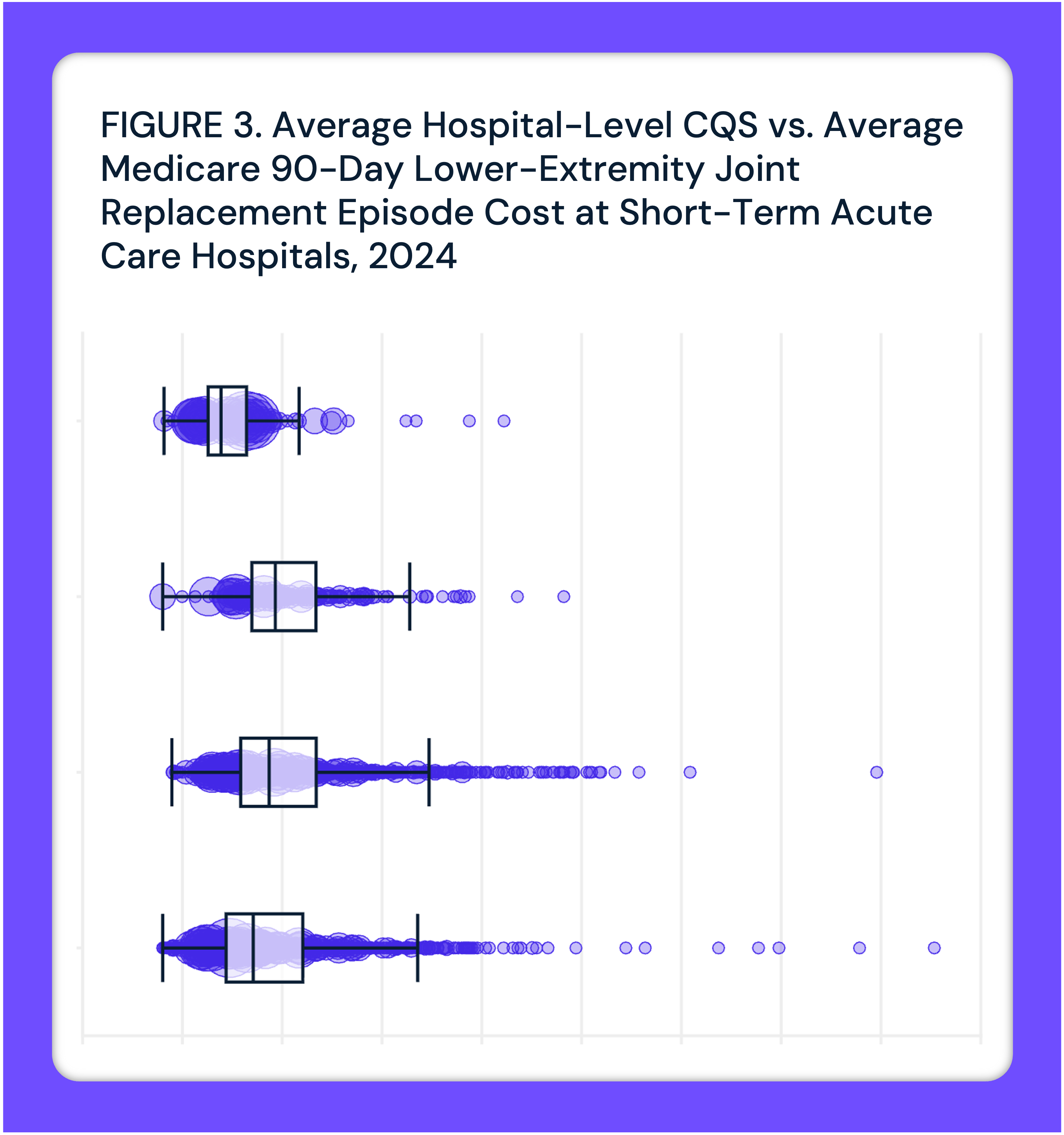
Episode Cost and Quality Are Not Correlated Across Hospitals Subject to Mandatory LEJR Bundled Payment Under TEAM and Proposed CJR-X
Read the StudyJune 04, 2026
Episode Cost and Quality Are Not Correlated Across Hospitals Subject to Mandatory LEJR Bundled Payment Under TEAM and Proposed CJR-X
There is no observable correlation (r=-0.1) between LEJR episode cost and quality across hospitals subject to TEAM and the proposed CJR-X model.
Katie Patton
StudiesMay 28, 2026
Pre-Diagnostic Symptom Patterns Preceding Colorectal and Ovarian Cancer Differ by Age as Early-Onset Incidence Increases
Early-onset colorectal and ovarian cancer incidence is increasing, yet younger patients face longer diagnostic delays due to nonspecific symptoms and lower clinical suspicion.
Katie Patton
StudiesMay 21, 2026
Behavioral Health Polypharmacy Is Growing Most Among Young Adults, With Disproportionate Growth in Stimulant Co-Prescribing
Behavioral health polypharmacy is rising fastest among adults 18–44, with stimulant co-prescribing growing sharply and women driving multi-class prescribing trends.
Katie Patton
StudiesMay 14, 2026
EVALI Encounters Remain Above 2020 Baseline as Vaping Prevalence Among Adolescents and Young Adults Grows
EVALI, a serious lung injury linked to vaping, with cases remaining above pre-2020 levels as e-cigarette use rises – predominantly affects young adults, especially women.
Katie Patton
StudiesMay 07, 2026
Nearly 80% of CBSAs Are Highly Concentrated for Both Inpatient Care and Commercial Health Insurance
In 2024, 76.8% of metro health markets were highly concentrated for both inpatient care and commercial insurance. Most markets see balanced power, but overall competition is limited nationwide.
Katie Patton
StudiesApril 30, 2026
The Majority of Hospital and Health Plan Reported Rates Diverge by More Than 10%
Less than half of hospital and health plan reported rates align with 10%; over 80% differ by more than 10%, with some diverging by over 17x, undermining price transparency and benchmarking.
Katie Patton
StudiesApril 23, 2026
As the Number of Residency Positions Increase in 2026, Primary Care Fill Rates Decline and Workforce Gaps Widen
2026 Match data shows increasing residency positions, but worsening gaps as primary care goes unfilled, signaling supply growth misaligned with workforce demand.
Clara Petrucelli
FundamentalsApril 21, 2026
The Demographic Composition of Local Markets Determines Localized Healthcare Demand
Demographic shifts – aging, declining births, slowing immigration – are reshaping U.S. healthcare demand unevenly across markets, demanding local analysis over national averages.
Clara Petrucelli
StudiesApril 16, 2026
Despite Behavioral Health Prescription Volume Growth, the Behavioral Health Treatment Gap Has Widened
Despite growing behavioral health prescriptions, the treatment gap widened – more patients go untreated (22.4% in 2024), stimulant use increased and allied health professionals now lead prescribing.
Katie Patton






















.png)

















.png?width=171&height=239&name=2025%20Trends%20Report%20Nav%20(1).png)
